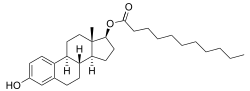
十一酸雌二醇
 | |
 | |
| 臨床資料 | |
|---|---|
| 读音 | shí yī suān cí èr chún(漢語拼音) |
| 商品名 | Delestrec, Progynon Depot 100, 以及其他 |
| 其他名稱 | EU;E2U; 雌二醇十一酸酯;十一酸雌酮;十一烷酸雌二醇 (商品代號)RS-1047;SQ-9993 |
| 给药途径 | 肌注[1] |
| 藥物類別 | 雌二醇; 雌二醇酯 |
| ATC碼 | |
| 法律規範狀態 | |
| 法律規範 |
|
| 藥物動力學數據 | |
| 生物利用度 | 肌注(IM ):接近100%[2] 口服:同口服雌激素药物,约为5%[3][4] |
| 血漿蛋白結合率 | Estradiol: ~98% (到白蛋白和SHBG)[2][5] |
| 药物代谢 | 在肝臟、血液和組織中通過酯酶的裂解[6][4] |
| 代謝產物 | 雌二醇,十一酸,雌二醇代謝物[6][4] |
| 生物半衰期 | 不確定 |
| 作用時間 | 肌注(IM ): • 10–12.5 mg: 1–2 月[7][8] • 25–50 mg: 2–4 月[9] |
| 排泄途徑 | 尿液 |
| 识别信息 | |
| |
| CAS号 | 3571-53-7 |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.020.616 |
| 化学信息 | |
| 化学式 | C29H44O3 |
| 摩尔质量 | 440.67 g·mol−1 |
| 3D模型(JSmol) | |
| |
| |
十一酸雌二醇(estradiol undecylate)縮寫EU或E2U,又稱十一酸雌酮,曾在Delestrec和Progynon Depot 100等品牌名下銷售,是一種雌激素藥物,曾用於治療男性攝護腺癌[10][11][12][13][1]。它還作為跨性別女性激素療法的一種方式[14][15][16]。儘管十一酸雌二醇過去曾經使用,但目前已停產[12][17]。該藥物通常攝入方式是每月一次的肌肉注射[1][18][13]。
十一酸雌二醇对男性的副作用可能包括乳房压痛、乳房发育、女性化、性功能障碍、不孕不育、液体滞留和心血管问题[18]。十一烷基雌二醇是一种雌激素,因此,是雌激素受体的激动剂,而雌激素受体是雌激素的生物靶标,如雌二醇[6][19][20]。它是一种雌激素酯,是体内雌二醇的一种非常持久[6]的前体药物[4]。正因为如此,它被认为是一种天然和生物相同的雌激素形式。注射十一酸雌二醇的持续时间约为1至4个月[8][9][7][21]。
十一酸雌二醇首次被描述是在1953年,並在1956年被引入醫學應用[8][22][9][23]。它一直在2000年代後期之前被使用,然後停產。十一酸雌二醇曾在歐洲市場上銷售,但似乎從未在美國上市[24][12][25]。多年來,它被用作治療男性攝護腺癌的肌注雌激素,儘管使用頻率不如聚磷酸雌二醇。有的跨性別女性會自行製作此種藥物以實施女性化激素療法。
医疗用途
[编辑]十一酸雌二醇曾被用作高剂量雌激素疗法的一种形式,用于治疗前列腺(俗稱“高雌抗雄”),但后来在这个适应症上被新一代药物取代,这些新药物具有更少的不良效应(例如乳腺增生和心血管并发症),如GnRH类似物和非甾体抗雄激素[1][26]。它已在许多临床研究中用于评估这个目的[27][28][29][30][31]。它以每3到4周(或每月一次)100毫克的剂量通过肌肉注射用于这个适应症[18][32]。
十一酸雌二醇有抗雄激素類似的效果,曾被用于抑制性犯罪者的性欲[33]。對此适应症,它的剂量为每3到4周一次,肌肉注射50到100毫克[33]。
十一酸雌二醇还曾用于治疗女性的乳腺癌[34]。它也在更年期激素疗法中使用,例如用于缓解潮热和其他更年期症状[9]。与雌二醇戊酸酯、雌二醇环戊酸酯和苯甲酸雌二醇一起,十一酸雌二醇已被用作肌肉注射雌激素,用于跨性別女性的激素疗法[14][15][16],剂量为每月100毫克至800毫克不等[15][16][14][35][36][37]。
可用形式
[编辑]十一酸雌二醇可作为肌肉注射的油溶液,一種規格是以100 mg/mL的藥物浓度的安瓿瓶。[38][39]
禁忌症
[编辑]十一酸雌二醇作為一種雌激素,禁忌症包括凝血问题、心血管疾病、肝脏疾病以及某些激素敏感性癌症,例如乳腺癌和子宫内膜癌等。[40][41][42][43]
副作用
[编辑]十一酸雌二醇及其副作用曾在由美因茨大學大學泌尿科Jacobi及其同事主持的III期國際多中心隨機對照試驗中進行評估,用於治療晚期前列腺癌[18][44][45][46][47][28][48]。該研究包括來自12個治療中心的191名患者,這些患者接受了為期6個月的肌肉注射,其中96名患者每月接受100毫克的十一酸雌二醇,另有95名患者每週接受300毫克的醋酸環丙孕酮[44][46][47][28][48][49][50]。該研究的一個分支首次於1978年和1980年報告了Mainz大學中心的42名患者[51][18][28][48]。
這些患者年齡在51到84歲之間(平均68歲),排除了有預先存在心血管疾病的患者[13][18][52]。對於十一酸雌二醇組報告了相當高的心血管併發症發生率(76%;21/16總發生率),其中心血管發病率為67%(21/14),心血管死亡率為9.5%(21/2)[13][18][52]。該組中的心血管發病包括外周水腫和表層靜脈炎(38%;21/8),冠心病(24%;21/5)和深靜脈血栓形成(4.8%;21/1)[18][52],而心血管死亡則包括心肌梗死(4.8%;21/1)和肺栓塞(4.8%;21/1)。
十一酸雌二醇組中的八例心血管併發症,包括兩例死亡,被認為是“嚴重的”[52][53]。相反,在环丙烯酸乙酯對照組中未發生心血管毒性(0%;21/0)[13][18][52]。十一酸雌二醇的其他副作用還包括乳腺增生(100%;21/21)和勃起功能障礙(90%;21/19)[18]。在這相對較小的研究中,十一酸雌二醇的心血管併發症與高劑量的聚磷酸雌二醇和經皮雌二醇治療前列腺癌的大規模和高質量的臨床研究形成對比,這些研究中觀察到最少或沒有心血管有害性[54][55][56][57][58]。
藥物过量
[编辑]十一酸雌二醇曾在臨床上以每月高達800至2400毫克的大劑量通過肌肉注射使用,分為每週兩到三次注射[59][60][61][62][63],每次注射100至200毫克。作為比較,有報告指出,單次100毫克肌肉注射的十一酸雌二醇可導致約500pg/mL的雌二醇水平[64]。雌激素過量的症狀可能包括噁心、嘔吐、腹脹、體重增加、水腫、乳房疼痛、陰道分泌物增加、腿沉和腿部抽筋[65]。這些副作用可以通過降低攝入劑量來減輕[65]。
互动
[编辑]药理
[编辑]药效学
[编辑]药代动力学
[编辑]化学性質
[编辑]历史
[编辑]社会与文化
[编辑]通用名
[编辑]雌二醇十一酸酯(Estradiol undecylate)是该药物的通用名称、国际非专利药品名称(INN)和美国药典通用名称(USAN)。[66][67][68][69] 有些出版物中也将其拼写为雌二醇十一酸酯,同时也被称为雌二醇十一酸酯。[70][66][67][68][69] 在德国,它有多种拼写,包括 estradiolundecylat、östradiolundecylat、östradiolundezylat、oestradiolundecylat、oestradiolundezylat 等。[71] 雌二醇十一酸酯还以其先前的开发代码名称 RS-1047 和 SQ-9993 而闻名。[66][67][68][69]
商品名
[编辑]雌二醇十一酸酯的主要品牌名称是 Progynon Depot 100。[66][67][68]除了 Progynon Depot 100 外,雌二醇十一酸酯还以其他品牌名称上市,包括 Delestrec、Depogin、Estrolent、Oestradiol D、Oestradiol-Retard Theramex 和 Primogyn Depot [0.1 mg/ml] 等。[66][67][68][72][73]
可用性
[编辑]雌二醇十一酸酯在欧洲(包括法国、德国、英国、摩纳哥、荷兰、瑞士)以及日本是可用的。[68][72][74][75][76] 然而,它已被停产,因此不再可获得。[77][78]
參考
[编辑]- ^ 1.0 1.1 1.2 1.3 Zink C. Dictionary of Obstetrics and Gynecology. Walter de Gruyter. 1 January 1988: 85. ISBN 978-3-11-085727-6.
- ^ 2.0 2.1 Stanczyk FZ, Archer DF, Bhavnani BR. Ethinyl estradiol and 17β-estradiol in combined oral contraceptives: pharmacokinetics, pharmacodynamics and risk assessment. Contraception. June 2013, 87 (6): 706–727. PMID 23375353. doi:10.1016/j.contraception.2012.12.011.
- ^ Emmens CW. Hormone Assay. Elsevier Science. 22 October 2013: 394–. ISBN 978-1-4832-7286-3.
- ^ 4.0 4.1 4.2 4.3 Kuhl H. Pharmacology of estrogens and progestogens: influence of different routes of administration. Climacteric. August 2005, 8 (Suppl 1): 3–63. PMID 16112947. S2CID 24616324. doi:10.1080/13697130500148875.
- ^ Falcone T, Hurd WW. Clinical Reproductive Medicine and Surgery. Elsevier Health Sciences. 2007: 22,362,388 [2024-01-18]. ISBN 978-0-323-03309-1. (原始内容存档于2023-01-10).
- ^ 6.0 6.1 6.2 6.3 Oettel M, Schillinger E. Estrogens and Antiestrogens II: Pharmacology and Clinical Application of Estrogens and Antiestrogen. Springer Science & Business Media. 6 December 2012: 261,544. ISBN 978-3-642-60107-1.
Natural estrogens considered here include: [...] Esters of 17β-estradiol, such as estradiol valerate, estradiol benzoate and estradiol cypionate. Esterification aims at either better absorption after oral administration or a sustained release from the depot after intramuscular administration. During absorption, the esters are cleaved by endogenous esterases and the pharmacologically active 17β-estradiol is released; therefore, the esters are considered as natural estrogens.
- ^ 7.0 7.1 引用错误:没有为名为
Labhart2012的参考文献提供内容 - ^ 8.0 8.1 8.2 引用错误:没有为名为
pmid13142295c的参考文献提供内容 - ^ 9.0 9.1 9.2 9.3 引用错误:没有为名为
pmid13391788的参考文献提供内容 - ^ Elks J. The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. 14 November 2014: 898–. ISBN 978-1-4757-2085-3.
- ^ Roberts AD. Dictionary of Steroids: Chemical Data, Structures, and Bibliographies. CRC Press. 1991: 415 [20 May 2012]. ISBN 978-0-412-27060-4.
- ^ 12.0 12.1 12.2 Index Nominum 2000: International Drug Directory. Taylor & Francis US. 2000: 405 [20 May 2012]. ISBN 978-3-88763-075-1.
- ^ 13.0 13.1 13.2 13.3 13.4 Norman G, Dean ME, Langley RE, Hodges ZC, Ritchie G, Parmar MK, et al. Parenteral oestrogen in the treatment of prostate cancer: a systematic review. British Journal of Cancer. February 2008, 98 (4): 697–707. PMC 2259178
 . PMID 18268497. doi:10.1038/sj.bjc.6604230.
. PMID 18268497. doi:10.1038/sj.bjc.6604230.
- ^ 14.0 14.1 14.2 Schlatterer K, von Werder K, Stalla GK. Multistep treatment concept of transsexual patients. Experimental and Clinical Endocrinology & Diabetes. 1996, 104 (6): 413–419. PMID 9021341. S2CID 25099676. doi:10.1055/s-0029-1211479.
- ^ 15.0 15.1 15.2 Benjamin H, Lal GB, Green R, Masters RE. The Transsexual Phenomenon. Ace Publishing Company. 1966: 107.
Another preparation of even higher potency is Squibb's Delestrec, which at this writing is not yet on the market in the United States, but is well known in Germany and other European countries under the name of Progynon Depot (Schering). It is chemically Estradiol Undecylate in oil, likewise slowly absorbing, and containing 100 mg. to 1 cc. Injections of 1 cc. once or twice a month can be sufficient. Occasionally, however, larger doses are required to influence the patient's emotional distress.
- ^ 16.0 16.1 16.2 Israel GE. Transgender Care: Recommended Guidelines, Practical Information, and Personal Accounts. Temple University Press. March 2001: 64–. ISBN 978-1-56639-852-7.
- ^ Estradiol. [2024-01-18]. (原始内容存档于2018-06-18).
- ^ 18.00 18.01 18.02 18.03 18.04 18.05 18.06 18.07 18.08 18.09 Jacobi GH, Altwein JE, Kurth KH, Basting R, Hohenfellner R. Treatment of advanced prostatic cancer with parenteral cyproterone acetate: a phase III randomised trial. British Journal of Urology. June 1980, 52 (3): 208–215. PMID 7000222. doi:10.1111/j.1464-410x.1980.tb02961.x.
- ^ 引用错误:没有为名为
Urology1973的参考文献提供内容 - ^ Lembeck F, Sewing KF. Pharmakologie-Fibel: Tafeln zur Pharmakologie-Vorlesung. Springer-Verlag. 7 March 2013: 113–. ISBN 978-3-642-65621-7.
- ^ 引用错误:没有为名为
WildeCoombs1959的参考文献提供内容 - ^ 引用错误:没有为名为
pmid13345960的参考文献提供内容 - ^ 引用错误:没有为名为
Bishop1958的参考文献提供内容 - ^ 引用错误:没有为名为
KleemannEngel2014的参考文献提供内容 - ^ 引用错误:没有为名为
IndexNominum2004的参考文献提供内容 - ^ Mutschler E, Derendorf H. Drug Actions: Basic Principles and Therapeutic Aspects. CRC Press. 1995: 609 [30 January 2013]. ISBN 978-0-8493-7774-7.
- ^ Enfedjieff M. [Experiences with hormonal treatment of prostatic carcinoma] [Experiences with hormonal treatment of prostatic carcinoma]. Zeitschrift für Urologie und Nephrologie. March 1974, 67 (3): 171–173. PMID 4848715. (原始内容存档于2018-11-23) (德语).
Treatment of prostatic carcinoma in 256 patients using parenteral injections of Progynon Depot (a depto estradiol preparation) is reproted. 58% of patients survived 3 or more years from beginning of treatment, and in 70% therapeutic results were considered good with regression of tumor mass, reduction or disappearance of pain, normalization of miction, and improved general status. Results of estrogen treatment are evident within 3 months in most cases. Side effects include gynecomastia in 95% of cases, impotence in almost all patients, and atrophic changes in the testicles, which may actually be desirable. Prostatectomy is not recommended because of the high incidence of metastases even when prostatic disease is still small, because of the high operative mortality, and because of the undesirable after-effects. Orchidectomy was performed in patients in whom the prostatic capsule had been invaded, or who had distant metastases. Estrogen therapy for prostatic carcinoma gives excellent results, and is very easy for both patient and physician.
- ^ 28.0 28.1 28.2 28.3 Tunn UW. Antiandrogene in der Therapie des fortgeschrittenen Prostatakarzinoms [Antiandrogens in the Treatment of Advanced Prostate Cancer]. Konservative Therapie des Prostatakarzinoms [Conservative Therapy of Prostate Cancer]. 1987: 113–121. ISBN 978-3-540-17724-1. doi:10.1007/978-3-642-72613-2_12 (德语).
- ^ Saborowski KJ, Konservative Therapie mit Cyproteronacetat und Estradiolundecylat beim Fortgeschrittenen Prostatacarcinom: Eine 5-Jahres-Studie [Conservative Therapy with Cyproterone Acetate and Estradiol Undecylate in Advanced Prostate Cancer: A 5-Year Study], Bochum, Univ., Diss., 1988, OCLC 917571781, OL 24895092W (德语)
- ^ Mollard P. [Clinical action of estradiol undecylate in the treatment of prostatic cancer] [Clinical action of estradiol undecylate in the treatment of prostatic cancer]. Lyon Medical. March 1963, 209: 759–765. PMID 13935867 (法语).
- ^ Schubert GE, Ziegler H, Völter D. [Comparison of histological and cytological studies of the prostate with special reference to oestrogene induced changes (author's transl)] [Comparison of histological and cytological studies of the prostate with special reference to oestrogene induced changes]. Verhandlungen der Deutschen Gesellschaft für Pathologie. 1973, 57: 315–318 [2018-11-23]. PMID 4142204. (原始内容存档于2018-11-23) (德语).
- ^ Satoskar RS, Bhandarkar SD, Rege NN. Pharmacology and Pharmacotherapeutics. Popular Prakashan. 1973: 934–. ISBN 978-81-7991-527-1.
- ^ 33.0 33.1 Morgan HG, Morgan MH. Aids to Psychiatry
 . Churchill Livingstone. 1984: 75. ISBN 978-0-443-02613-3.
. Churchill Livingstone. 1984: 75. ISBN 978-0-443-02613-3. Treatment of sexual offenders. Hormone therapy. [...] Oestrogens may cause breast hypertrophy, testicular atrophy, osteoporosis (oral ethinyl oestradiol 0.01-0.05 mg/day causes least nausea). Depot preparation: oestradiol [undecyleate] 50-100mg once every 3–4 weeks. Benperidol or butyrophenone and the antiandrogen cyproterone acetate also used.
- ^ Kennedy BJ. Effect of massive doses of estradiol undecylate in advanced breast cancer. Cancer Chemother Rep. April 1967, 51 (2): 491–495.
- ^ 引用错误:没有为名为
pmid2978262的参考文献提供内容 - ^ 引用错误:没有为名为
pmid2528051的参考文献提供内容 - ^ 引用错误:没有为名为
pmid18057351的参考文献提供内容 - ^ Kleemann A, Engel J, Kutscher B, Reichert D. Pharmaceutical Substances: Syntheses, Patents and Applications of the most relevant APIs 5th. Thieme. 2009: 1167–1174. ISBN 978-3-13-179525-0.
- ^ 引用错误:没有为名为
Martindale的参考文献提供内容 - ^ 引用错误:没有为名为
pmid2215269的参考文献提供内容 - ^ Lauritzen C, Studd JW. Current Management of the Menopause. CRC Press. 22 June 2005: 95–98,488. ISBN 978-0-203-48612-2.
- ^ Laurtizen C. Hormone Substitution Before, During and After Menopause (PDF). Fisch FH (编). Menopause – Andropause: Hormone Replacement Therapy Through the Ages. Krause & Pachernegg: Gablitz. 2001: 67–88 [2024-01-18]. ISBN 978-3-901299-34-6. (原始内容存档 (PDF)于2018-05-10).
- ^ Midwinter A. Contraindications to estrogen therapy and management of the menopausal syndrome in these cases. Campbell S (编). The Management of the Menopause & Post-Menopausal Years: The Proceedings of the International Symposium held in London 24–26 November 1975 Arranged by the Institute of Obstetrics and Gynaecology, The University of London. MTP Press Limited. 1976: 377–382. ISBN 978-94-011-6167-1. doi:10.1007/978-94-011-6165-7_33.
- ^ 44.0 44.1 Jacobi GH. Intramuscular cyproterone acetate treatment for advanced prostatic carcinoma: results of the first multicentric randomized trial. Schröder FH (编). Proceedings Androgens and Anti-androgens, International Symposium, Utrecht, June 5th, 1982. Schering Nederland BV. June 1982: 161–169. ISBN 978-9090004327. OCLC 11786945.
- ^ Tunn UW, Graff J, Senge T. Treatment of inoperable prostatic cancer with cyproterone acetate. Schröder FH (编). Proceedings Androgens and Anti-androgens, International Symposium, Utrecht, June 5th, 1982. Schering Nederland BV. June 1982: 149–159. ISBN 978-9090004327. OCLC 11786945.
- ^ 46.0 46.1 Tunn UW, Radlmaier A, Neumann F. Antiandrogens in Cancer Treatment. Stoll BA (编). Endocrine Management of Cancer: Contemporary Therapy. 1988: 43–56. ISBN 978-3-8055-4686-7. doi:10.1159/000415355.
- ^ 47.0 47.1 Schröder FH, Radlmaier A. Steroidal Antiandrogens. Jordan VC, Furr BJ (编). Hormone Therapy in Breast and Prostate Cancer. Cancer Drug Discovery and Development. Humana Press. 2009: 325–346. ISBN 978-1-60761-471-5. doi:10.1007/978-1-59259-152-7_15.
- ^ 48.0 48.1 48.2 Ackermann R, Altwein JE, Faul P. Aktuelle Therapie des Prostatakarzinoms. Springer-Verlag. 13 March 2013: 276–277. ISBN 978-3-642-84264-1.
- ^ Namer M. Clinical applications of antiandrogens. Journal of Steroid Biochemistry. October 1988, 31 (4B): 719–729. PMID 2462132. doi:10.1016/0022-4731(88)90023-4.
- ^ Jacobi GR, Tunn UW, Senge TH. Clinical experience with cyproterone acetate for palliation of inoperable prostate cancer. Jacobi GH, Hohenfellner R (编). Prostate Cancer. Williams & Wilkins. 1 December 1982: 305–319. ISBN 978-0-683-04354-9.
- ^ Altwein JE, Jacobi GH, Hohenfellner R. Estrogen versus cyproterone acetate in untreated inoperable carcinoma of the prostate: first results of an open, prospective, randomized study. Abstracts 3rd Congress of the European Association of Urology, Monte Carlo. 1978.
- ^ 52.0 52.1 52.2 52.3 52.4 Saygin D, Tabib T, Bittar HE, Valenzi E, Sembrat J, Chan SY, et al. Transcriptional profiling of lung cell populations in idiopathic pulmonary arterial hypertension. Pulmonary Circulation. 1983, 10 (1): 40–48. PMC 7052475
 . PMID 32166015. S2CID 23447326. doi:10.1007/BF00326861.
. PMID 32166015. S2CID 23447326. doi:10.1007/BF00326861.
- ^ Jacobi GH, Wenderoth UK. Gonadotropin-releasing hormone analogues for prostate cancer: untoward side effects of high-dose regimens acquire a therapeutical dimension. European Urology. 1982, 8 (3): 129–134. PMID 6281023. doi:10.1159/000473499.
- ^ Ockrim J, Lalani EN, Abel P. Therapy Insight: parenteral estrogen treatment for prostate cancer--a new dawn for an old therapy. Nature Clinical Practice. Oncology. October 2006, 3 (10): 552–563. PMID 17019433. S2CID 6847203. doi:10.1038/ncponc0602.
- ^ Lycette JL, Bland LB, Garzotto M, Beer TM. Parenteral estrogens for prostate cancer: can a new route of administration overcome old toxicities?. Clinical Genitourinary Cancer. December 2006, 5 (3): 198–205. PMID 17239273. doi:10.3816/CGC.2006.n.037.
- ^ Russell N, Cheung A, Grossmann M. Estradiol for the mitigation of adverse effects of androgen deprivation therapy. Endocrine-Related Cancer. August 2017, 24 (8): R297–R313. PMID 28667081. doi:10.1530/ERC-17-0153
 .
.
- ^ Langley RE, Cafferty FH, Alhasso AA, Rosen SD, Sundaram SK, Freeman SC, et al. Cardiovascular outcomes in patients with locally advanced and metastatic prostate cancer treated with luteinising-hormone-releasing-hormone agonists or transdermal oestrogen: the randomised, phase 2 MRC PATCH trial (PR09). The Lancet. Oncology. April 2013, 14 (4): 306–316. PMC 3620898
 . PMID 23465742. doi:10.1016/S1470-2045(13)70025-1.
. PMID 23465742. doi:10.1016/S1470-2045(13)70025-1.
- ^ Langley RE, Gilbert DC, Duong T, Clarke NW, Nankivell M, Rosen SD, et al. Transdermal oestradiol for androgen suppression in prostate cancer: long-term cardiovascular outcomes from the randomised Prostate Adenocarcinoma Transcutaneous Hormone (PATCH) trial programme (PDF). Lancet. February 2021, 397 (10274): 581–591 [2024-01-18]. PMC 7614681
 . PMID 33581820. S2CID 231885186. doi:10.1016/S0140-6736(21)00100-8. (原始内容存档 (PDF)于2024-01-18).
. PMID 33581820. S2CID 231885186. doi:10.1016/S0140-6736(21)00100-8. (原始内容存档 (PDF)于2024-01-18).
- ^ 引用错误:没有为名为
Kennedy19672的参考文献提供内容 - ^ 引用错误:没有为名为
Israel20012的参考文献提供内容 - ^ Asscheman H, Gooren LJ, Assies J, Smits JP, de Slegte R. Prolactin levels and pituitary enlargement in hormone-treated male-to-female transsexuals. Clinical Endocrinology. June 1988, 28 (6): 583–588. PMID 2978262. S2CID 29214187. doi:10.1111/j.1365-2265.1988.tb03849.x.
- ^ Asscheman H, Gooren LJ, Eklund PL. Mortality and morbidity in transsexual patients with cross-gender hormone treatment. Metabolism. September 1989, 38 (9): 869–873. PMID 2528051. doi:10.1016/0026-0495(89)90233-3.
- ^ Gazzeri R, Galarza M, Gazzeri G. Growth of a meningioma in a transsexual patient after estrogen-progestin therapy. The New England Journal of Medicine. December 2007, 357 (23): 2411–2412. PMID 18057351. doi:10.1056/NEJMc071938
 .
.
- ^ 引用错误:没有为名为
pmid1231448的参考文献提供内容 - ^ 65.0 65.1 Lauritzen C. Clinical use of oestrogens and progestogens. Maturitas. September 1990, 12 (3): 199–214. PMID 2215269. doi:10.1016/0378-5122(90)90004-P.
- ^ 66.0 66.1 66.2 66.3 66.4 引用错误:没有为名为
Elks20142的参考文献提供内容 - ^ 67.0 67.1 67.2 67.3 67.4 引用错误:没有为名为
Roberts19912的参考文献提供内容 - ^ 68.0 68.1 68.2 68.3 68.4 68.5 引用错误:没有为名为
IndexNominum20002的参考文献提供内容 - ^ 69.0 69.1 69.2 引用错误:没有为名为
Drugs.com82的参考文献提供内容 - ^ 引用错误:没有为名为
pmid12314482的参考文献提供内容 - ^ von Bruchhausen F, Dannhardt G, Ebel S, Frahm AW, Hackenthal E, Holzgrabe U. Hagers Handbuch der Pharmazeutischen Praxis: Band 8: Stoffe E-O. Springer-Verlag. 2 July 2013: 84–. ISBN 978-3-642-57994-3.
- ^ 72.0 72.1 引用错误:没有为名为
KleemannEngel20143的参考文献提供内容 - ^ Index Nominum: International Drug Directory. CRC Press. 2004: 469–. ISBN 978-3-88763-101-7.
- ^ Boschann HW. Observations of the role of progestational agents in human gynecologic disorders and pregnancy complications. Annals of the New York Academy of Sciences. July 1958, 71 (5): 727–752. Bibcode:1958NYASA..71..727B. PMID 13583829. doi:10.1111/j.1749-6632.1958.tb46803.x.
- ^ Bishop PM. Endocrine Treatment of Gynaecological Disorders. Gardiner-Hill H (编). Modern Trends in Endocrinology 1. London: Butterworth & Co. 1958: 231–244.
- ^ Bishop PM. Chemistry of the Sex Hormones. Thomas. 1962: 78.
- ^ Micromedex. [2024-02-04]. (原始内容存档于2018-09-17).
- ^ Sweetman SC (编). Sex hormones and their modulators. Martindale: The Complete Drug Reference 36th. London: Pharmaceutical Press. 2009: 2098. ISBN 978-0-85369-840-1.
